Nanomaterials show promise for atheroprotective regenerative immunotherapy
CRN Principal Investigator: Evan Scott, Associate Professor of Biomedical Engineering and of Microbiology-Immunology
Cardiovascular disease (CVD) is the leading cause of death in the developed world and is responsible for over 17% of national health care expenditures in the United States. With 45% of the US population projected to experience some form of CVD by 2035, the annual costs are expected to reach $1.1 trillion. New strategies for preventing and treating CVD are desperately needed. A principle cause of CVD is atherosclerosis, which is marked by the accumulation of fat-laden plaques (i.e. atheromas) within the walls of arterial vessels. Rupture of these plaques can induce heart attacks, usually without warning, so we need methods for both early detection and regeneration of these unstable arterial lesions.
A Catalyst Award from the Center for Regenerative Nanomedicine (CRN) has allowed my laboratory to develop a novel nanotechnology-based method to address these issues. Through the first publication from my newly formed lab (Yi et al. ACS Nano, 2016), we demonstrated that altering the size, shape and structure of nanomaterials can significantly enhance their targeted delivery of imaging agents and therapeutics to critical components of the immune system that contribute to atherosclerosis. We have filed a patent application for this method, which we refer to as Nanostructure Enhanced Targeting (NSET). NSET is a key component of our ongoing work to develop clinically relevant and safe strategies to both detect unstable atheromas and treat CVD using immunotherapy (Allen et al. Nano Research, 2018).
Seed funding from our CRN Catalyst project helped us develop targeted nanomaterials to validate our untested, high-risk idea, which was subsequently awarded a five-year, $2.3 million New Innovator Award from the National Institutes of Health. By combining our NSET methodology with targeting peptides specific for atheroma-resident immune cells, we developed novel nanocarriers to transport diverse anti-inflammatory therapeutics, like vitamin D3 or celastrol (Allen et al. Biomaterials Science, 2019), to inhibit vascular plaque development. We tested these nanocarriers in transgenic mouse models of atherosclerosis and found that they significantly inhibited plaque formation and prevented stiffening of arteries (Yi et al. Advanced Functional Materials, 2019). Encouraged by these results, research into cardiovascular immunotherapy has become a key focus of my lab, resulting in a recent publication demonstrating a sustained delivery system for nanomaterials that can modulate vascular inflammation for weeks following a single injection (Yi et al. Frontiers in Bioengineering and Biotechnology, 2020). We are extremely grateful for the early stage funding and continued support from the CRN.
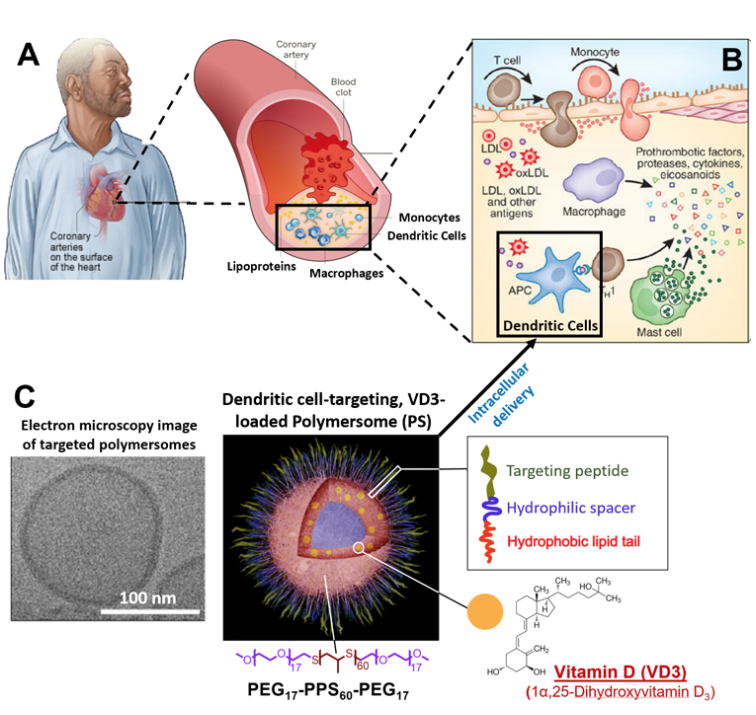
Anti-inflammatory nanocarriers inhibit dendritic cell-derived plaque inflammation. (A) The coronary artery is highly susceptible to a dangerous accumulation of lipid within the vessel wall when exposed to a high-fat diet. Inflammation greatly increases the chances of these vascular lesions (i.e. atheromas) rupturing, which can result in a heart attack or stroke. (B) Inflammation within plaques is driven by inflammatory molecules released by dendritic cells when they are exposed to oxidized low-density lipoprotein (oxLDL). (C) Schematic of the dendritic cell-targeting poly(ethylene glycol)-block-poly(propylene sulfide) (PEG-b-PPS) polymersome (PS). These nanocarriers are decorated with a custom targeting peptide construct, which was optimized for intracellular delivery to dendritic cells. Inhibition of inflammatory pathways within dendritic cells is mediated by vitamin D (1α,25-Dihydroxyvitamin D3), which is a potent anti-inflammatory loaded within the PSs. *From Yi et al. Advanced Functional Materials, 2019.
